WASHINGTON – Thousands of insulin pumps are being recalled as the FDA warns of a hacking risk.
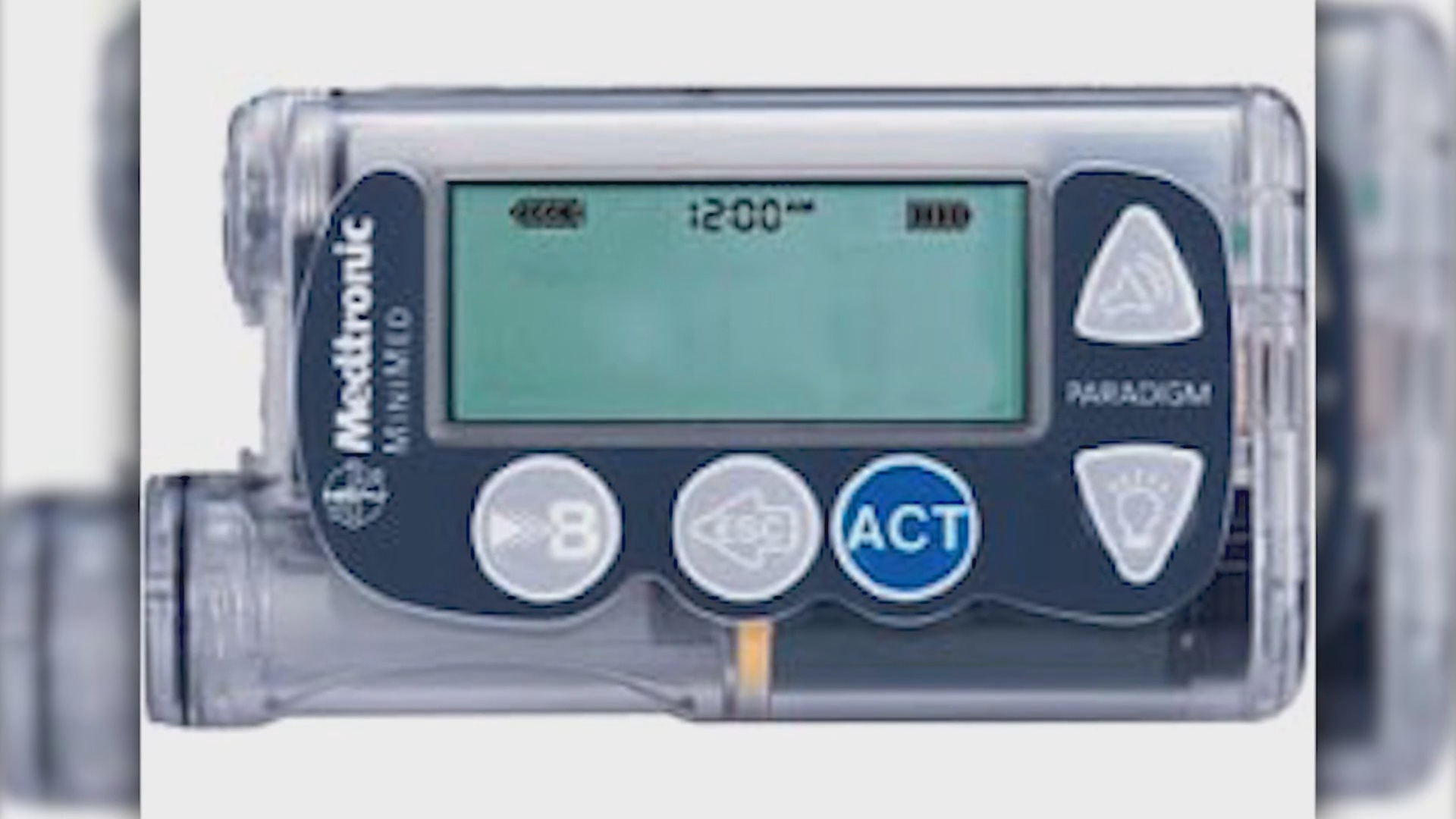
Medtronic is recalling its Mini-Med Paradigm Series Insulin Pumps and its Mini-Med 508 Insulin Pump.
Insulin pumps are small computerized devices that deliver insulin doses to diabetics.
The FDA warns someone nearby could connect wirelessly and then potentially hack into the devices, change the settings, and deliver too much or too little insulin.
The FDA said it’s not aware of anyone actually doing this; but if it’s not addressed, they say the risk to patients “significant.”
Medtronic said it’s identified about four-thousand patients who may be using pumps that are vulnerable to the issue and is working to determine if there are more.
Medtronic is providing alternative insulin pumps to patients that have enhanced built-in cybersecurity features.




Leave a Reply