WASHINGTON – A recall for blood pressure drugs has been expanded.
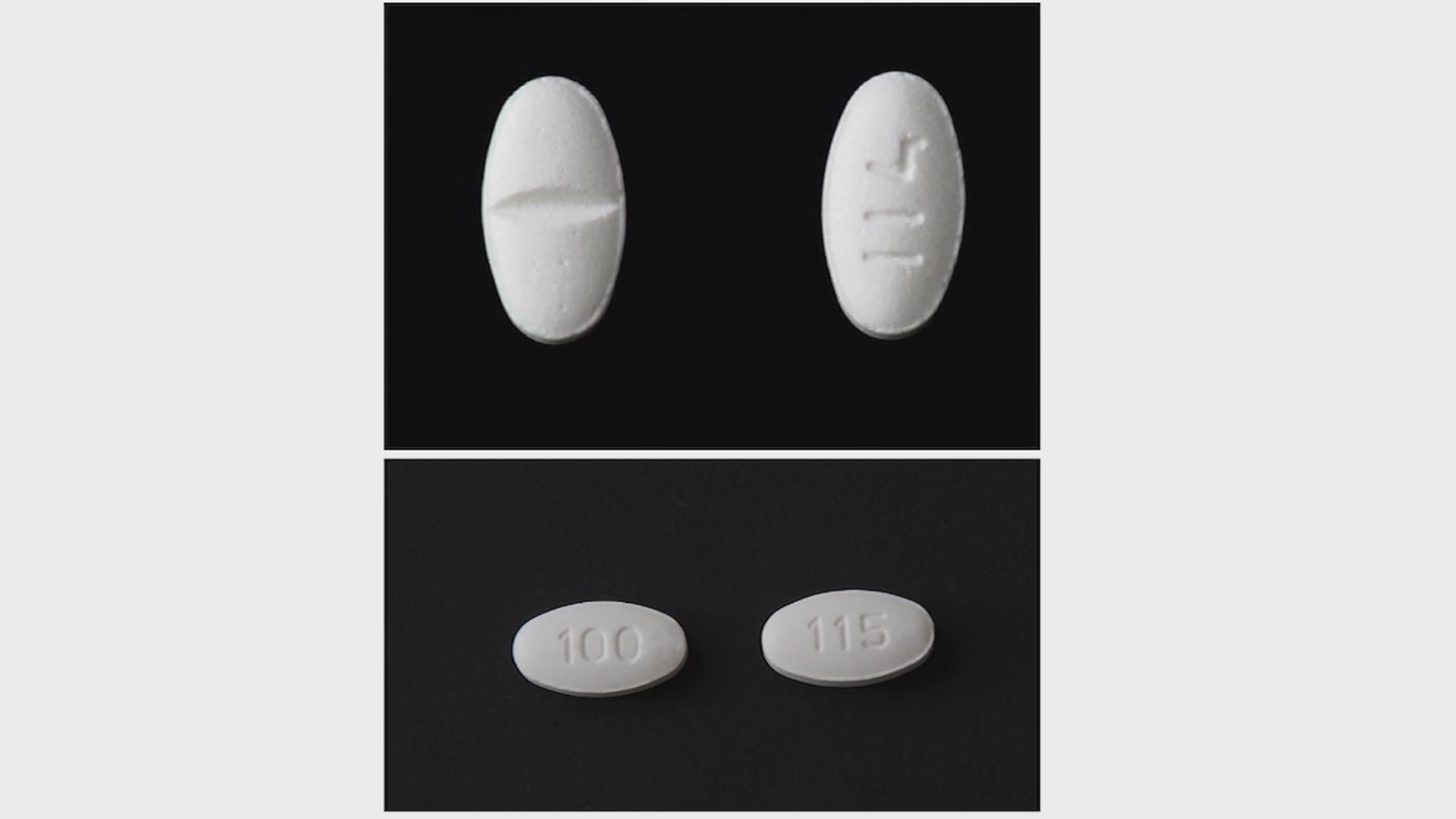
Torrent Pharmaceuticals says two of its blood pressure medications were found to have trace amounts of a potentially cancer-causing impurity.
The voluntary recall affects Losartan Potassium Tablets USP and Losartan Potassium-Hydrochlorothiazide Tablets.
This recall is related to the Valsartan recall that has been expanded multiple times since July, when separate lots of blood pressure medications from different companies were pulled from pharmacy shelves.
The medications all contained either Valsartan, Losartan or Irbesartan and had traces of these impurities that pose a cancer risk to users.




Leave a Reply