App users, tap here to watch video.
NEW YORK – A New York State hospital has administered the first COVID-19 vaccination in the United States.
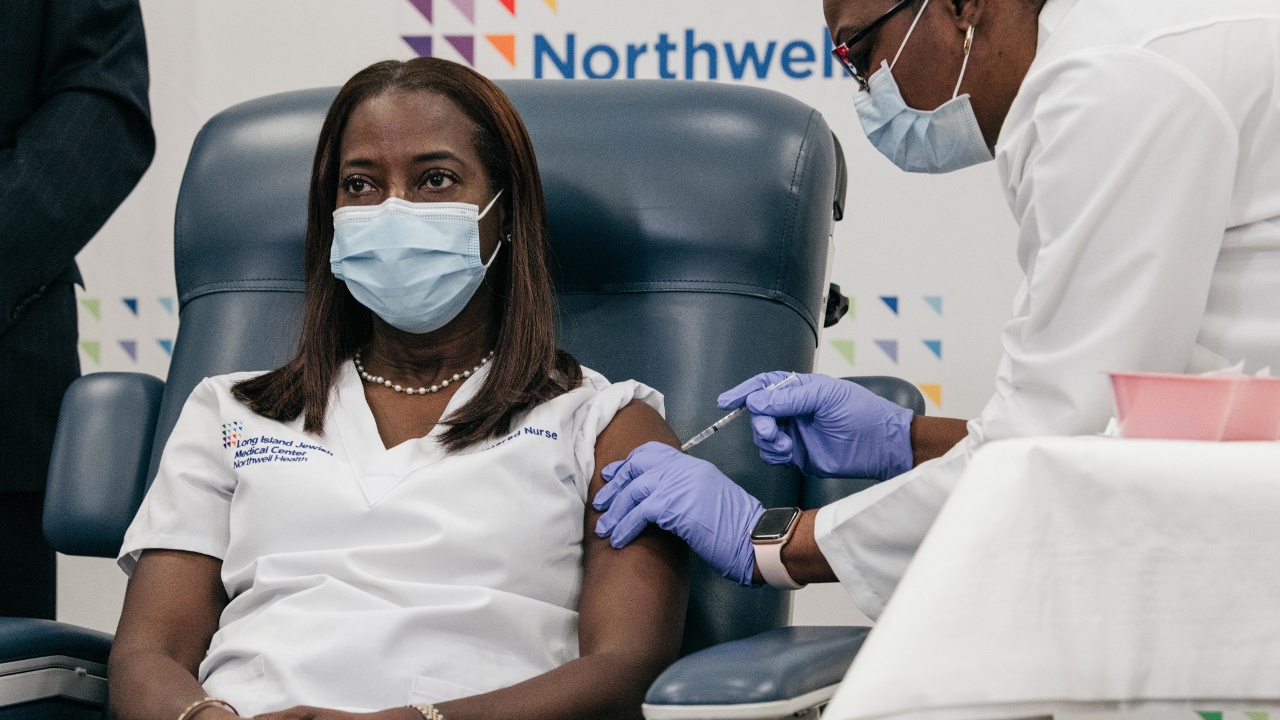
Sandra Lindsay, a critical care nurse, received the first dose at Northwell Health on Monday morning in New York City.
“I want to instill confidence that the vaccine is safe,” she said after receiving the shot.
The vaccination was livestreamed by New York State Governor Andrew Cuomo, who says the vaccine only works if Americans take it.

“It is going to take months before the vaccine hits critical mass, so this is the light at the end of the tunnel, but, it is a long tunnel,” said Cuomo speaking remotely via Zoom. “We need people to do the right thing though the holiday season, hopefully by June we can hit critical mass.”
The vaccine is administered in two doses, with the second given 29 days after the first.
Cuomo thanked the nurse who was vaccinated and other healthcare workers across the state.
The vaccine was given the ‘go ahead’ to be administered in the United States over the weekend.
In a statement, CDC Director Robert Redfield says the official recommendation follows last week’s FDA decision to authorize emergency use of the vaccine.
“Last night, I was proud to sign the Advisory Committee on Immunization Practices’ recommendation to use Pfizer’s COVID-19 vaccine in people 16 and older,” said Redfield in a statement. “As COVID-19 cases continue to surge throughout the U.S., CDC’s recommendation comes at a critical time.”
The CDC says healthcare personnel and long-term care facility residents should be vaccinated first.
The Advisory Committee on Immunization Practices voted Saturday to recommend the Pfizer vaccine for ages 16 and up.
The U.S. Food and Drug Administration authorized the first COVID-19 vaccine developed by Pfizer Inc and BioNTech SE Friday, making it available for emergency use to patients aged 16 and older.
In clinical trials, the vaccine was 95% effective at preventing illness and showed no short-term safety issues, officials say.
The first trucks carrying the vaccine left a Michigan manufacturing plant earlier Sunday.
Shipments of the Pfizer vaccine set in motion the biggest vaccination effort in American history at a critical juncture of the pandemic that has killed 1.6 million and sickened 71 million worldwide according to data complied by Johns Hopkins University.




Leave a Reply